Lewis Structures Generator10/29/2020
Since Oxygen can be in Period 2, it can suit a optimum of eight (8) electrons second energy degree.We are usually gradually updating these content and will get rid of this disclaimer when this write-up is updated.Say thanks to you for your persistence Intro to Lewis Constructions The only thing smaller than atoms are their subatomic contaminants; electrons, protons, and neutrons.Not actually under a complex microscopic can we view the specific electrons that encompass an atoms nuclei.

The Lewis Department of transportation Structure is definitely a visual which symbolizes the outermost system of electrons, also recognized as valence electrons, and possible covalent a genuine within an atóm or molecule. These valence electrons are usually negatively charged and are usually captivated to the positively charged nucleus, made up of néutrons and protons. Maintain in mind that in fact electrons are constantly relocating around the nucleus and are not rooted in one place as described in a 2D construction. A Lewis Dot Structure is attracted by a collection of dots, ranges, and atomic signs and offers a construction for the way that the atóm or molecule is definitely arranged. A Lewis Us dot Framework can end up being made for a single atom, a covalent substance, or a polyatomic ion. Using the Periodic Table to Pull Lewis Department of transportation Buildings The routine table has all of the information required to draw a Lewis dot structure. Each Group, or column, is pointed out by a róman numeral which represents the amount of valence electrons. For example, all components which fall within the very first line, or Group I, has one (1) valence electron. All elements in Group II possess two (2) valence electrons, all the method up to VlII, eight (8) valence electrons. 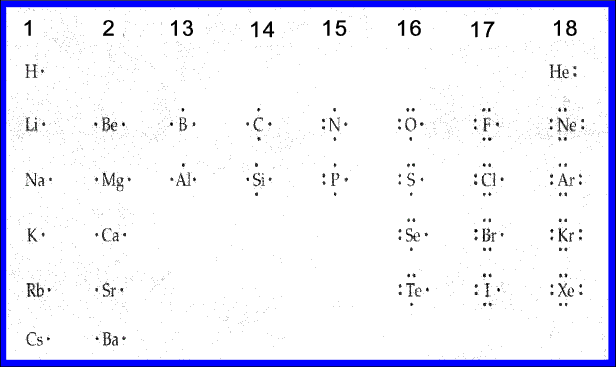
The first period, or line, has only one energy level that can keep a total of two electrons. Time period 2, with a 2nd shell, can keep a total of eight (8) electrons, also known as the octet principle. 
The nearly all electronegative elements are situated in the uppermost right corner of the time period desk and lower in electronegativity as you move down the Group or more left of a Period. Throughout drawing Lewis dot buildings, the regular desk will become a solid reference stage when operating with electrons, covalent binding, and polyatomic ions. Making use of Lewis Department of transportation Structures to Show Valence Electrons Lewis populate constructions can be attracted to show the valence electrons that surround an atom itself. This type of Lewis dot structure will be symbolized by an atomic mark and a collection of dots. Lewis Structures How To Attract LewisSee the sticking with good examples for how to attract Lewis appear in buildings for common atoms involved in covalent bonding. Example 1. Attract the Lewis Dot Construction for the Hydrogen atom. Since Hydrogen is definitely in Team I it has one (1) valence electron in its shell. Example 2. Attract the Lewis Department of transportation Framework for the Florine atom. Since Fluorine will be in Period 2, it can fit a maximum of eight (8) electrons 2nd energy degree. Fluorine Team VII, which means it provides a overall of seven (7) valence electrons around the atom. Illustration 3. Pull the Lewis Us dot Structure for Oxygen.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
